Nitric Acid (HNO3)
![]()
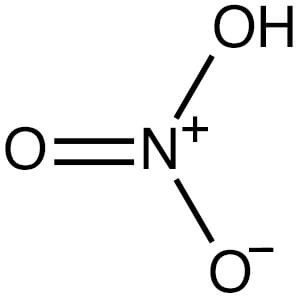
Nitric acid (HNO3, CAS 7697-37-2) is a highly toxic mineral acid also known as aqua fortis or spirit of niter. It is colorless, but can turn pale yellow when exposed to air. Solutions that contain more than 86% nitric acid are referred to as “fuming nitric acid.”
Nitric acid is typically available as an azeotrope (68%) or as a reagent (70%). In the laboratory, it is used in the synthesis of nitro compounds and as an analytical reagent, a strong oxidizing agent, and a dye intermediate.
In industry, nitric acid is used to manufacture inks, dyes, and nitrogenous fertilizers; produce plastics and polymers; and create explosives like TNT and nitroglycerine.
Nitric acid is highly corrosive in nature, so personal protective equipment (PPE) should be used to reduce harmful exposure to the acid and its vapors. Risks include inhalation, ingestion, and absorption, with possible severe damage to eyes, skin, and mucous membranes.
Store nitric acid away from incompatible materials, including organic compounds, metals, alcohols, and bases.